Accueil du site > Production scientifique > Globule to Helix Transition in Sodiated Polyalanines
Globule to Helix Transition in Sodiated Polyalanines
Date de publication: 26 octobre 2012
J. K. Martens, I. Compagnon, E. Nicol, T. B. McMahon, C. Clavaguéra, G. Ohanessian
J. Phys. Chem. Lett. 3 (22) 3320–3324 (2012). DOI
Travail réalisé sur le site de l’Université Paris-Sud.
Abstract
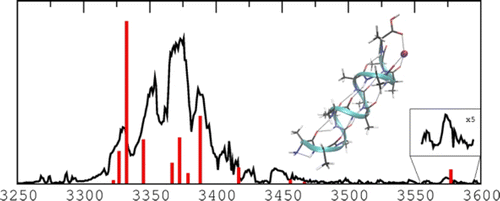
The structures of sodiated polyalanine peptides containing 8–12 residues are investigated using infrared multiple photon dissociation (IRMPD) spectroscopy and classical and quantum modeling. Calculations indicate that the α-helical structure is the most stable conformation for the peptides whatever their size. The IRMPD spectra provide evidence for the coexistence of helical and globular shapes for Ala8Na+, and possibly for Ala9Na+. The turning point from globule to helix is thus found at Ala8–9Na+. The N–H and O–H stretching region allows identifying a new spectroscopic pattern typical for alpha-helical structures of polyalanines.








