Accueil du site > Production scientifique > Tyrosine nitration as evidenced by IRMPD spectroscopy
Tyrosine nitration as evidenced by IRMPD spectroscopy
Date de publication: 1er décembre 2011
R. K. Sinha, B. Chiavarino, M. E. Crestoni, D. Scuderi, S. Fornarini
Int J Mass Spectrom Ion Processes 308 (2-3) 209-216 (2011). DOI
Travail réalisé sur le site de l’Université Paris Sud.
Abstract
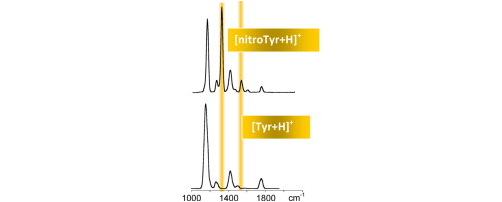
Protonated 3-nitrotyrosine ([nitroTyr + H]+) has been investigated by collision induced dissociation at variable energy and by IR multiple photon dissociation (IRMPD) spectroscopy in conjunction with quantum chemical calculations. Ultimately, this investigation is aimed at providing a diagnostic signature of protein tyrosine nitration, a post translational modification implied in pathological states. IRMPD spectroscopy of [nitroTyr + H]+ ions has been examined in two different spectral regions, namely in the 1000–2000 cm-1 range, using the free electron laser beamline at the CLIO facility, and in the 3200–3700 cm-1 range, using a tabletop laser source. [nitroTyr + H]+ ions have been assayed in parallel with intact [Tyr + H]+ ions revealing characteristic bands at 1329 and 1540 cm-1. Mode assignments allowed by density functional theory calculations of the IR spectra of the most stable conformers indicate that these features are associated to vibrations of the nitro group and are endowed with enhanced activity. In the NH/OH stretching region, the remarkable difference between the IRMPD spectra is the strong absorption at 3641 cm-1 of the ring OH stretching of [Tyr + H]+ which is absent in the nitrated ion due to a strong hydrogen bond engaging the ring OH and the ortho nitro group. These results suggest that IRMPD spectroscopy and tandem mass spectrometry may afford a selective method for the analysis and characterization of 3-nitrotyrosine, to be possibly extended to 3-nitrotyrosine-containing peptides.








