Accueil du site > Production scientifique > Structure of singly hydrated, protonated phospho-tyrosine
Structure of singly hydrated, protonated phospho-tyrosine
Date de publication: 1er décembre 2011
D. Scuderi, J. M. Bakker, S. Durand, P. Maitre, A. Sharma, J. K. Martens, E. Nicol, C. Clavaguéra, G. Ohanessian
Int J Mass Spectrom Ion Processes 308 (2-3) 338-347 (2011). DOI
Travail réalisé sur le site de l’Université Paris Sud.
Abstract
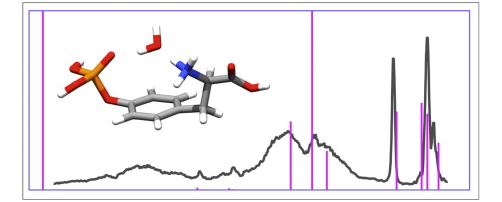
Tandem mass spectrometry is a powerful analytical technique for distinguishing phosphorylated from non-phosphorylated peptides, and also to locate the phosphorylated site. It is however desirable to further extend the tandem MS capacities via the development of structure specific activation techniques. This work is part of an ongoing project which aims at characterizing the environment of the phosphate group using tunable IRMPD. The effect of microsolvation of protonated phospho-tyrosine on the phosphate environment has been investigated using a combination of tandem MS experiments and quantum chemical calculations. Infrared spectra have been recorded in the 900–1900 cm-1 and 2500–3750 cm-1 regions using a free electron laser and a tabletop laser, respectively. Water is found to form a hydrogen bonded bridge between the otherwise nearly isolated phosphate and ammonium groups. Theoretical and experimental results also provide consistent evidences for two weaker hydrogen bonds between the two other NH bonds with the π-aromatic ring and the carboxylic CO. The MP2 calculated IR absorption spectrum of the lowest energy structure allows for a clear band assignment. As compared to intramolecular direct interaction between the phosphate and the ammonium, the two hydrogen bonds associated with the water bridge between these groups are found to have a larger impact on their IR structure-specific probes. This is reflected by the red-shift of the P=O stretch, and also the blue shift of the umbrella mode of the ammonium. Special attention is paid to the 2500–3750 cm-1 spectral range. While the phosphate OH stretching modes are not affected by the addition of water, the red shifts of the three ammonium NH stretches are found to be significant and quite different for their three types of non-covalent interactions.








