Accueil du site > Production scientifique > Experimental and Computational Investigation of Salophen-Zn Gas Phase Complexes with Cations : A Source of Possible Interference in Anionic Recognition
Experimental and Computational Investigation of Salophen-Zn Gas Phase Complexes with Cations : A Source of Possible Interference in Anionic Recognition
Date de publication: 30 août 2017
A. Ciavardini ; S. Fornarini ; A. Dalla Cort ; S. Piccirillo ; D. Scuderi ; E. Bodo
J. Phys. Chem. A 121(37) 7042-7050 (2017). DOI
Travail réalisé sur le site de l’Université Paris Sud.
Abstract
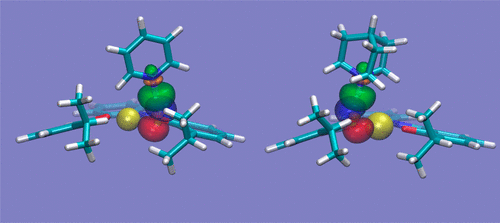
We explore the possibility that protonated molecular ions might be an unexpected source of interference in the recognition process of anions and neutral species by Zn-salophen receptors. Zn-salophen complexes are known to bind anions and neutral molecules in solution. We present here evidence (from computational work and IRMPD spectroscopy) that these complexes can also be the binding site for protonated pyridine or quinuclidine. The resulting binding pattern does not involve the Zn ion, but one of the oxygen atoms directly attached to it. The resulting complex therefore turns out to have a positive charge adjacent to the Zn-salophen binding site. This finding seems to point to the existence of an interfering factor in the quantification of the experimental data about the association constant.








