Accueil du site > Production scientifique > Electron detachment/photodetachment dissociation of lasso peptides
Electron detachment/photodetachment dissociation of lasso peptides
Date de publication: 15 novembre 2015
M. Perot-Taillandier, C. Afonso, Q. Enjalbert, R. Antoine, P. Dugourd, R. B. Cole, J.-C. Tabet, S. Rebuffat
Int. J. Mass Spectrom. 390 91-100 (2015). DOI
Travail réalisé sur le site de l’Université Pierre et Marie Curie.
Abstract
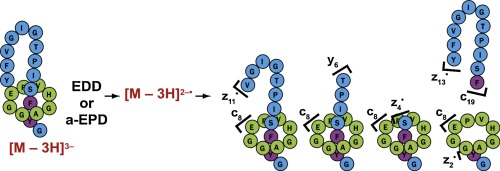
Lasso peptides are bioactive peptides produced by bacteria, characterized by a mechanically interlocked topology where the C-terminal tail of the peptide is threaded through and trapped within an N-terminal macrolactam ring. The structural characterization of lasso peptides and differentiation from their unthreaded topoisomers by mass spectrometry are challenging tasks. We previously explored the fragmentation mechanisms of lasso peptides in positive ion mode and showed several signatures of the lasso topology under collision induced dissociation (CID) and electron capture dissociation. Here we analyzed the dissociation of the multiply-deprotonated microcin J25 (MccJ25), a 21-residue lasso peptide produced by Escherichia coli, together with its non-lasso topoisomer and several variants generated by site-directed mutagenesis, under different modes of activation. The fragmentation patterns obtained by CID for the threaded and unthreaded structures were very similar. By contrast, electron detachment dissociation (EDD) as well as activated-electron photodetachment dissociation (a-EPD) revealed very different dissociation pathways for the two topoisomers. The doubly deprotonated topoisomers showed a different deprotonation pattern, Tyr20 residue being deprotonated for MccJ25 only, yielding the singly charged [c19−C2H4O] product ion. MccJ25 also triggered several two-peptide product ions diagnostic of the lasso topology, including the [(c8)*(z2)]








