Accueil du site > Production scientifique > Structure of Pb2+/dCMP and Pb2+//CMP complexes as characterized by tandem mass spectrometry and IRMPD spectroscopy
Structure of Pb2+/dCMP and Pb2+//CMP complexes as characterized by tandem mass spectrometry and IRMPD spectroscopy
Date de publication: 1er juillet 2011
J.-Y. Salpin, L. Gamiette, J. Tortajada, T. Besson, P. Maître
Int J Mass Spectrom Ion Processes 304 (2011) 154– 164 (2011). DOI
Travail réalisé sur le site de l’Université Paris Sud.
Abstract
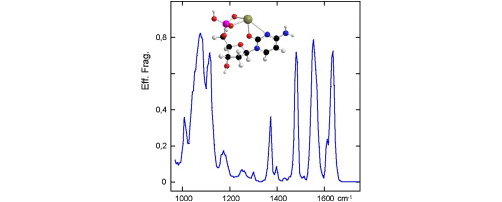
The structure of Pb2+/cytidine-5′-monophosphate (CMP) and Pb2+/deoxycytidine-5′-monophosphate (dCMP) complexes were probed in the gas phase by combining electrospray ionization, tandem mass spectrometry and mid-infrared multiple-photon dissociation (IRMPD) spectroscopy. The fragment ions detected upon collision suggest that the metal interacts with both the phosphate group and the cytosine. This finding is further confirmed by IRMPD spectroscopy. The IRMPD spectrum of the ESI-generated [Pb(dCMP)−H]+ and [Pb(CMP)−H]+ complexes is indeed in very good agreement with DFT-computed infrared absorption spectra of macrochelate forms, in which the Pb2+ ion not only interacts with the phosphate group but also with the carbonyl group of the nucleobase moiety, by folding of the mononucleotides. The structures thus characterized therefore differ from those proposed in solution and deduced from potentiometric studies. Our data also suggest that losing the nucleobase residue under CID conditions does not necessarily mean a lack of interaction between the metal and the nucleobase moiety, as commonly mentioned in the literature for large oligonucleotides.








